Latanoprostene bunod
Today we want to talk about Latanoprostene bunod, a topic that has generated interest and controversy over time. Latanoprostene bunod is a matter that has been talked about in different areas and that has generated conflicting opinions. Throughout history, Latanoprostene bunod has been the object of study, debate and analysis, since its relevance and impact are undeniable. In this article we will explore different aspects related to Latanoprostene bunod, from its origin to its influence today. In addition, we will seek to understand the importance of Latanoprostene bunod and how it has marked a before and after in various contexts. Without a doubt, Latanoprostene bunod is a topic that does not leave anyone indifferent and that will continue to generate interest in the future.
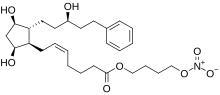 | |
| Clinical data | |
|---|---|
| Trade names | Vyzulta |
| Other names | BOL-303259-X |
| AHFS/Drugs.com | Multum Consumer Information |
| License data | |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.251.571 |
| Chemical and physical data | |
| Formula | C27H41NO8 |
| Molar mass | 507.624 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Latanoprostene bunod (trade name Vyzulta) is an ophthalmic drug approved in the United States in 2017 for the reduction of intraocular pressure in patients with open-angle glaucoma or ocular hypertension. It targets the trabecular meshwork directly.
References
- ^ "Search Page - Drug and Health Product Register". 23 October 2014.
- ^ "Vyzulta- latanoprostene bunod solution/ drops". DailyMed. 1 May 2019. Retrieved 7 June 2022.
- ^ "FDA Approves Vyzulta (latanoprostene bunod) Ophthalmic Solution for Open-Angle Glaucoma, Ocular Hypertension" (Press release). Valeant Pharmaceuticals International, Inc.
- ^ a b Hoy SM (May 2018). "Latanoprostene Bunod Ophthalmic Solution 0.024%: A Review in Open-Angle Glaucoma and Ocular Hypertension". Drugs. 78 (7): 773–780. doi:10.1007/s40265-018-0914-6. PMC 5976683. PMID 29761382.